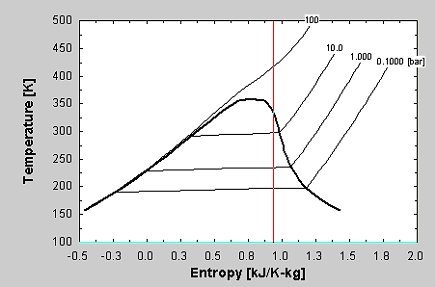
Since entropy is a state variable, just depending upon the beginning and end states, these expressions can be used for any two points that can be put on one of the standard graphs. Using the ideal gas lawīut since specific heats are related by C P = C V + R. and taking the entropy change upon binding into account if necessary/desired. The vibrational temperature of 'H'2 (the temperature at which it accesses its vibrational states) is Theta(vib) '6333 K'. Thus the calculation would take an inordinate amount of time to converge. This is a useful calculation form if the temperatures and volumes are known, but if you are working on a PV diagram it is preferable to have it expressed in those terms. How to calculate the change in the entropy of hydrogen molecule before and after the adsorption Chemistry Thermochemistry Entropy. 
Making use of the first law of thermodynamics and the nature of system work, this can be written To use this online calculator for Standard Entropy Change given Gibbs Free Energy, enter Change in Enthalpy (H), Gibbs Free Energy (G) & Temperature (T) and hit the calculate button. Add the product to the enthalpy difference to get the Gibbs energy free. 
Multiply the temperature change to get Entropy. The Enthalpy, temperature, and the Entropy of change can be viewed. With kT/2 of energy for each degree of freedom for each atom.įor processes with an ideal gas, the change in entropy can be calculated from the relationship You can easily calculate the Gibbs-free energy of chemical reactions by following the steps. This gives an expression for internal energy that is consistent with equipartition of energy. Determine the standard entropies of all products and reactants using the entropy table. This online calculator calculates information gain, the change in information entropy from a prior state to a state that takes some information as given. Then making use of the definition of temperature in terms of entropy: Expanding the entropy expression for V f and V i with log combination rules leads toįor determining other functions, it is useful to expand the entropy expression using the logarithm of products to separate the U and V dependence. One of the things which can be determined directly from this equation is the change in entropy during an isothermal expansion where N and U are constant (implying Q=W). The entropy S of a monoatomic ideal gas can be expressed in a famous equation called the Sackur-Tetrode equation. Versions 12 and higher have bromine-containing molecules.Entropy of an Ideal Gas Entropy of an Ideal Gas This free online devise entropy calculator is helpful to find the change in entropy for a reaction, change in Gibbs free energy and change in entropy of. 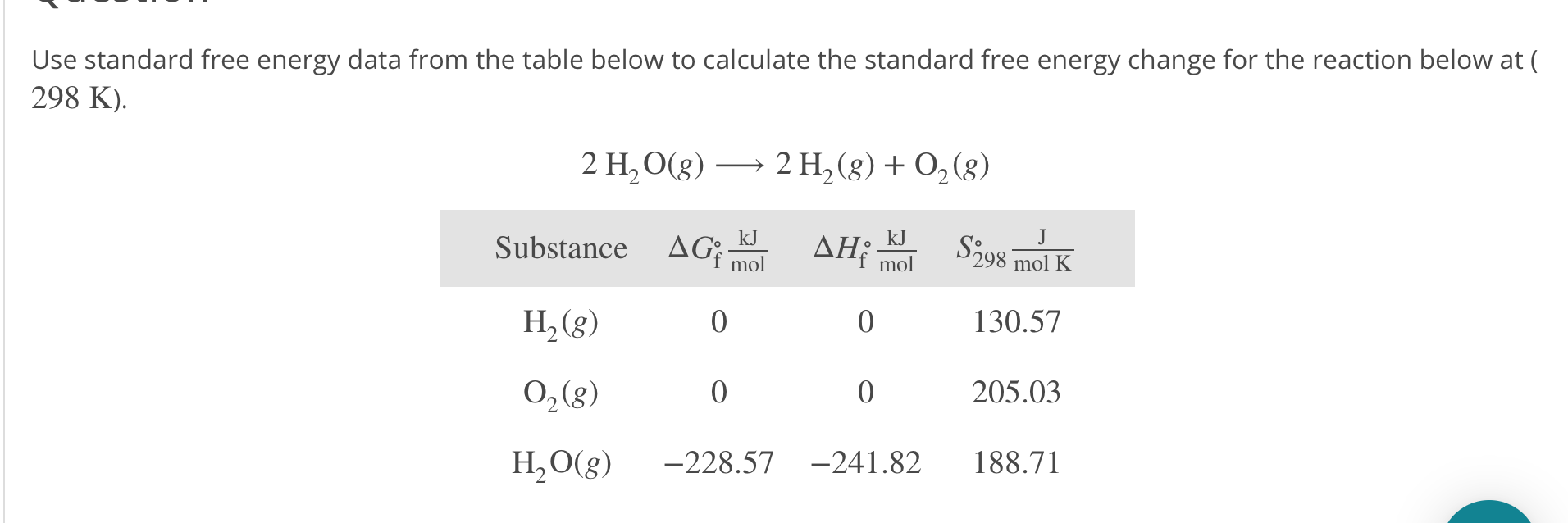
Versions 8 and higher have a few substituted benzenes with more than six heavy atoms. Six or fewer heavy atoms and twenty or fewer total atoms.See section I.B.1 for a periodic table view. Mostly atoms with atomic number less than than 36 (Krypton), except for most of the transition metals.Ions are indicated by placing + or - at the end of the formula (CH3+, BF4-, CO3-).Note that for this type of free energy calculation we solely need the. A comma delimited list of several species may be entered. First search for Free energy, then change init-lambda to 1 and change.Multiple specifications for an atom will be added.Parentheses may be used to group atoms.If only one of a given atom is desired, you may omit.Use our free online app Entropy Calculator for an ideal gas to determine all important calculations with parameters and constants. To specify the amounts of desired elements (e.g., C6H6). Find Entropy Calculator for an ideal gas at CalcTown. Enter a sequence of element symbols followed by numbers.Same old same old - this is just products minus reactants. You are here: OR Calculated > Entropy and Heat Capacity > Entropy OR Comparisons > Entropy > EntropyĬomparison of Entropies for a given moleculeĪt different levels of theory Select species by entering formula Rules for chemical formula 15.2 Calculate the standard entropy change for a reaction using standard entropy values.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
